
As leading remote patient monitoring vendors, we engineer the PM6100 for high-precision physiological tracking. Optimized for global medical distributors and healthcare facility integration.
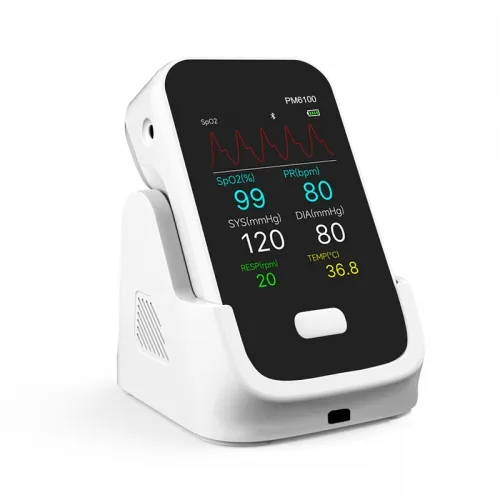
Features robust Bluetooth data transmission capabilities, ensuring stable integration with hospital HIS/EMR systems and dedicated medical applications.
Delivers clinical-grade accuracy across core metrics including SpO2, Non-Invasive Blood Pressure (NIBP), Pulse Rate (PR), and continuous ECG monitoring.
Designed with customizability in mind. Hardware interfaces, software algorithms, and external branding can be tailored to meet specific distributor requirements.

Engineered by top-tier remote patient monitoring devices manufacturers, the PM6100 delivers reliable, continuous vital signs tracking. Its compact architecture integrates seamlessly into diverse clinical workflows, ensuring rapid data transmission and optimal patient care across medical facilities.
Feedback from medical distributors and facility procurement teams.
"The PM6100's NIBP and SpO2 accuracy meets strict clinical requirements. We deployed 200 units across our regional clinics, and the wireless data transfer has significantly reduced manual charting errors."
"Finding a reliable OEM partner is tough. Berrymed provided exceptional flexibility with the PM6100, allowing us to customize the firmware and branding. Lead times were consistently met."
"As system integrators, the Bluetooth API stability of the PM6100 is outstanding. We seamlessly connected these monitors to our custom telehealth platform within weeks. Highly recommended."
Analysis of the evolving landscape in remote patient monitoring and the technological advancements shaping the industry.
Evaluating production capacity, clinical accuracy standards, and international certifications in OEM partnerships.
A strategic guide for procurement officers selecting vital signs monitors for diverse clinical environments.
For standard wholesale orders, our MOQ is designed to support both mid-sized clinics and large distributors. Specific MOQ requirements depend on whether you require standard configurations or customized OEM variations. Please contact our sales team for exact tier pricing.
Standard inventory shipments are typically processed within 7-14 business days. For OEM/ODM orders requiring customized firmware or branding, the lead time is generally 3-5 weeks following prototype approval.
The PM6100 is manufactured in our ISO 13485 certified facility and complies with stringent international medical device regulations, ensuring seamless import and distribution in major global markets.
Yes. We offer comprehensive OEM/ODM services including boot-logo modification, customized housing colors, personalized UI elements, and tailored packaging to align with your regional brand identity.
The PM6100 utilizes standardized Bluetooth protocols. We provide SDK/API documentation to qualified system integrators to facilitate secure and stable data transmission of physiological parameters directly into your proprietary HIS/EMR platforms.
The PM6100 utilizes advanced digital signal processing algorithms specifically optimized for complex clinical environments. It maintains high accuracy in physiological tracking even during low perfusion states (down to 0.1%) and motion artifact scenarios, ensuring reliable continuous monitoring.